Chondroitin Sulfate Sodium
Chondroitin,hydrogen sulfate,sodium salt [9082-07-9].
»Chondroitin Sulfate Sodium is the sodium salt of the sulfated linear glycosaminoglycan obtained from bovine,porcine,or avian cartilages of healthy and domestic animals used for food by humans.Chondroitin Sulfate Sodium consists mostly of the sodium salt of the sulfate ester of N-acetylchondrosamine (2-acetamido-2-deoxy-b-D-galactopyranose)and D-glucuronic acid copolymer.These hexoses are alternately linked b-1,4and b-1,3in the polymer.Chondrosamine moieties in the prevalent glycosaminoglycan are monosulfated primarily on position 4and less so on position 6.It contains not less than 90.0percent and not more than 105.0percent of chondroitin sulfate sodium,calculated on the dried basis.
NOTE—Chondroitin Sulfate Sodium is extremely hygroscopic once dried.Avoid exposure to the atmosphere,and weigh promptly.
Packaging and storage—
Preserve in tight containers.
Labeling—
Label it to indicate the species of the source from which the article was derived.
Clarity and color of solution—
Transfer 2.5g of Chondroitin Sulfate Sodium to a 50-mLvolumetric flask.Dissolve in and dilute with carbon dioxide-free water to volume,mix,and examine immediately.Measure the absorbance of this solution at 420nm in a 1-cm cell,using carbon dioxide-free water as the blank:its absorbance is not greater than 0.35.
Identification—
A:Infrared Absorption á197Kñ.
B:
Asolution containing 0.5g in 10mLof water meets the requirements of the test forSodium á191ñ.
Specific rotation á781Sñ:
between -20.0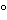 and -30.0
and -30.0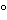 .
.
Test solution:
30mg per mL.
Microbial enumeration á2021ñ—
The total bacterial count does not exceed 1000cfu per g,and the total combined molds and yeasts count does not exceed cfu 100cfu per g.It meets the requirements of the tests for absence ofSalmonella species,andEscherichia coli.
Delete the following:
Test Preparation—
Provide separate 10-g specimens for each of the tests called for below.Dissolve Chondroitin Sulfate Sodium in pH7.2Phosphate buffer.[NOTE—On the basis of results for Preparatory Testing,modify the Test Preparationas appropriate.]
Preparatory Testing—
Incubate Clostridium sporogenes(ATCC No.11437)for 18to 24hours,and then dilute with pH7.2Phosphate Buffer.Inoculate the Test Preparation,to obtain a final concentration of less than 100cfu per mL.Controls containing the inoculum but without the material under test are prepared at the same time.Proceed as directed under Procedure,making sure to evaluate the growth after each time a medium is added.
Results—
Proceed as directed for Preparatory Testingunder Microbial Enumeration Tests—Nutritional and Dietary Supplements á2021ñ.
REINFORCED MEDIUM FOR CLOSTRIDIA
| Beef Extract | 10.0g |
| Peptone | 10.0g |
| Yeast Extract | 3.0g |
| Soluble Starch | 1.0g |
| Glucose Monohydrate | 5.0g |
| Cysteine Hydrochloride | 0.5g |
| Sodium Chloride | 5.0g |
| Sodium Acetate | 3.0g |
| Agar | 0.5g |
| Water | 1000mL |
Dissolve agar in water by heating to boiling,while stirring continuously.Adjust the pHif necessary,and sterilize.
pHafter sterilization:6.8±0.2.
COLUMBIA AGAR
| Pancreatic Digest of Casein | 10.0g |
| Meat Peptic digest | 5.0g |
| Heart Pancreatic digest | 3.0g |
| Yeast Extract | 5.0g |
| Corn Starch | 1.0g |
| Sodium Chloride | 5.0g |
| Agar | 15.0g |
| Water | 1000mL |
Dissolve agar in water by heating to boiling with continuous stirring.If necessary,adjust the pH.Sterilize,and allow to cool to 45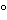 to 50
to 50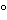 .Add gentamicin sulfate,equivalent to about 20mg of gentamicin base,and pour into Petri dishes.
.Add gentamicin sulfate,equivalent to about 20mg of gentamicin base,and pour into Petri dishes.
pHafter sterilization:7.3±0.2.
Procedure—
Take two equal portions of the Test Preparation,heat one to 80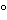 for 10minutes,and cool rapidly.Transfer 10mLof each portion to separate containers,each containing each 100mLof Reinforced Medium for Clostridia,and incubate under anaerobic conditions at 35
for 10minutes,and cool rapidly.Transfer 10mLof each portion to separate containers,each containing each 100mLof Reinforced Medium for Clostridia,and incubate under anaerobic conditions at 35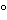 to 37
to 37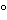 for 48hours.After incubation subculture each specimen on Columbia Agar Mediumto which gentamicin has been added,and incubate under anaerobic conditions at 35
for 48hours.After incubation subculture each specimen on Columbia Agar Mediumto which gentamicin has been added,and incubate under anaerobic conditions at 35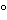 to 37
to 37 for 48hours.Examine the plates,and interpret as follows:if no growth of Gram-positive rods is detected,the test specimen meets the requirement for the absence of Clostridium species.
for 48hours.Examine the plates,and interpret as follows:if no growth of Gram-positive rods is detected,the test specimen meets the requirement for the absence of Clostridium species.
If growth occurs,subculture each distinct colony on Columbia Agar Medium,and separately incubate in aerobic and in anaerobic conditions at 35 to 37
to 37 for 48hours.The occurrence of only anaerobic growth of Gram-positive bacilli,giving a negative catalase reaction,indicates the presence of Clostridium sporogenes species.To perform the catalase test,transfer discrete colonies to glass slides,and apply a drop of dilute hydrogen peroxide solution:the reaction is negative if no gas bubbles evolve.If the test specimen exhibits none of these characteristics,it meets the requirement for the absence of Clostridiumspecies.
for 48hours.The occurrence of only anaerobic growth of Gram-positive bacilli,giving a negative catalase reaction,indicates the presence of Clostridium sporogenes species.To perform the catalase test,transfer discrete colonies to glass slides,and apply a drop of dilute hydrogen peroxide solution:the reaction is negative if no gas bubbles evolve.If the test specimen exhibits none of these characteristics,it meets the requirement for the absence of Clostridiumspecies. USP28
USP28
pHá791ñ:
between 5.5and 7.5,in a solution (1in 100).
Loss on drying á731ñ—
Dry it at 105 for 4hours:it loses not more than 10.0%of its weight.[NOTE—Chondroitin Sulfate Sodium is extremely hygroscopic once dried.Avoid exposure to the atmosphere,and weigh promptly.]
for 4hours:it loses not more than 10.0%of its weight.[NOTE—Chondroitin Sulfate Sodium is extremely hygroscopic once dried.Avoid exposure to the atmosphere,and weigh promptly.]
Residue on ignition á281ñ:
between 20.0%and 30.0%,on the dried basis.
Chloride á221ñ—
A0.10-g portion shows no more chloride than corresponds to 0.7mLof 0.020Nhydrochloric acid:not more than 0.50%is found.
Sulfate á221ñ—
Dissolve 200mg in 40mLof water.Add 10mLof a solution of cetylpyridinium chloride having a concentration of about 30mg per mL,mix,and pass through a filter.A25-mLportion of the filtrate shows no more sulfate than corresponds to 0.25mLof 0.020Nsulfuric acid:not more than 0.24%is found.
Heavy metals,Method IIá231ñ:
0.002%.
Organic volatile impurities,Method Iá467ñ:
meets the requirements.
Change to read:
Electrophoretic purity(see Electrophoresis á726ñ)—
Staining reagent:
 0.1%toluidine blue in acetic acid;dissolve 1g of toluidine blue in 1000mLof 0.1Macetic acid.
0.1%toluidine blue in acetic acid;dissolve 1g of toluidine blue in 1000mLof 0.1Macetic acid. USP28
USP28
Standard solution 1—
Prepare a solution of USP Chondroitin Sulfate Sodium RSin water having a known concentration of about  30mg per mL.
30mg per mL. USP28
USP28
Standard solution 2—
Dilute 1mLofStandard solution 1with water to  50mL,
50mL, USP28and mix.
USP28and mix.
Test solution—
Transfer 150mg of Chondroitin Sulfate Sodium,accurately weighed,to a  5.0-mL
5.0-mL USP28volumetric flask,dissolve in and dilute with water to volume,and mix.
USP28volumetric flask,dissolve in and dilute with water to volume,and mix.
Procedure—
 Fill the chambers of an electrophoresis apparatus suitable for separations on cellulose acetate membranes1(a small submarine gel chamber or one dedicated to membrane media)with 0.1M Barium acetate buffer,pH5.0.Soak a cellulose acetate membrane about 5to 6cm ×12to 14cm in 0.1M Barium acetate buffer,pH5.0for 10minutes,or until evenly wetted,then blot dry between two sheets of absorbent paper.Using an applicator2suitable for electrophoresis,apply equal volumes (about 0.5µL)of the Test solution,Standard solution 1,and Standard solution 2to the brighter side of the membrane held in position in an appropriate applicator stand or on a separating bridge in the chamber.Ensure that both ends of the membrane are dipped at least 0.5-to 1.0-cm deep into the buffer chambers.Apply a constant 60volts (about 6mAat the start)for 2hours.[NOTE—Perform the application of solutions and voltage within 5minutes because further drying of the blotted paper reduces sensitivity.]Place the membrane in a plastic staining tray,and with the application side down,float or gently immerse in Staining reagentfor 5minutes.Then stir the solution gently for 1minute.Remove the membrane,and destain in 5%acetic acid until the background clears.Compare the bands.The electropherogram obtained from the Test solutionexhibits a major band that is identical in position to the band obtained from Standard solution 1.The band obtained from Standard solution 2is clearly visible at a mobility similar to the band obtained from Standard solution 1.Any secondary band in the electropherogram of the Test solutionis not more intense than the band obtained from Standard solution 2.Not more than 2%of any individual impurity is found.Document the results by taking a picture within 15minutes of completion of destaining.
Fill the chambers of an electrophoresis apparatus suitable for separations on cellulose acetate membranes1(a small submarine gel chamber or one dedicated to membrane media)with 0.1M Barium acetate buffer,pH5.0.Soak a cellulose acetate membrane about 5to 6cm ×12to 14cm in 0.1M Barium acetate buffer,pH5.0for 10minutes,or until evenly wetted,then blot dry between two sheets of absorbent paper.Using an applicator2suitable for electrophoresis,apply equal volumes (about 0.5µL)of the Test solution,Standard solution 1,and Standard solution 2to the brighter side of the membrane held in position in an appropriate applicator stand or on a separating bridge in the chamber.Ensure that both ends of the membrane are dipped at least 0.5-to 1.0-cm deep into the buffer chambers.Apply a constant 60volts (about 6mAat the start)for 2hours.[NOTE—Perform the application of solutions and voltage within 5minutes because further drying of the blotted paper reduces sensitivity.]Place the membrane in a plastic staining tray,and with the application side down,float or gently immerse in Staining reagentfor 5minutes.Then stir the solution gently for 1minute.Remove the membrane,and destain in 5%acetic acid until the background clears.Compare the bands.The electropherogram obtained from the Test solutionexhibits a major band that is identical in position to the band obtained from Standard solution 1.The band obtained from Standard solution 2is clearly visible at a mobility similar to the band obtained from Standard solution 1.Any secondary band in the electropherogram of the Test solutionis not more intense than the band obtained from Standard solution 2.Not more than 2%of any individual impurity is found.Document the results by taking a picture within 15minutes of completion of destaining. USP28
USP28
Change to read:
Limit of protein—
Alkaline cupric tartaric reagent—
Dissolve 200mg of sodium tartrate dihydrate in 10mLof water,and mark as Solution A.Dissolve 100mg of cupric sulfate in 10mLof water,and mark as Solution B.Dissolve 2.0g of anhydrous sodium carbonate in 0.1Msodium hydroxide,dilute with 0.1Msodium hydroxide to 100mL,and mark as Solution C.Mix well 1mLof Solution Aand 1mLof Solution B,and to the mixture slowly add 100mLof Solution Cwith stirring.Use within 24hours,and discard afterwards.
Standard solution—
Transfer an accurately measured volume of 7percent bovine serum albumin certified standard to a suitable container,and dilute quantitatively and stepwise with water to obtain a solution having a known concentration of about 35µg per mL.
Test solution—
Transfer an accurately weighed quantity of Chondroitin Sulfate Sodium,equivalent to 60mg of the dried substance,to a 100-mLvolumetric flask,dissolve in and dilute with water to volume,and mix.
Procedure—
Add 2.0mLof freshly prepared Alkaline cupric tartaric reagentto test tubes containing 2.0mLof water,2.0mLof theTest solution,or 2.0mLof theStandard solution,and mix.After about 10minutes,add 1.0mLof Folin-Ciocalteu phenol TS, diluted with water (1:5)and
diluted with water (1:5)and USP28prepared immediately before use,to each test tube,and mix
USP28prepared immediately before use,to each test tube,and mix  immediately and vigorously.
immediately and vigorously. USP28After 30minutes,measure the absorbance of each solution at 750nm against the blank.The absorbance of theTest solution is not greater than the absorbance of theStandard solution:not more than 6.0%of proteins is found,calculated on the dried basis.
USP28After 30minutes,measure the absorbance of each solution at 750nm against the blank.The absorbance of theTest solution is not greater than the absorbance of theStandard solution:not more than 6.0%of proteins is found,calculated on the dried basis.
Change to read:
Content of chondroitin sulfate sodium—
Cetylpyridinium chloride solution—
Prepare a solution of cetylpyridinium chloride in water having a concentration of about 1mg per mL. Degas before use.
Degas before use.
Diluent—
Weigh about 297mg of monobasic potassium phosphate,492mg of dibasic potassium phosphate,and 250mg of polysorbate 80,and transfer into a 1-Lbeaker.Dissolve in 1000mLof water,and adjust with potassium hydroxide or phosphoric acid to a pHof 7.0±0.2. USP28
USP28
Standard solutions—
 Prepare a solution having a known concentration of USP Chondroitin Sulfate Sodium RSin water,
Prepare a solution having a known concentration of USP Chondroitin Sulfate Sodium RSin water, USP28and dilute with water,quantitatively and stepwise if necessary,to obtain threeStandard solutionshaving known concentrations of about
USP28and dilute with water,quantitatively and stepwise if necessary,to obtain threeStandard solutionshaving known concentrations of about  1.5mg per mL,1.0mg per mL,and 0.5mg per mL,respectively.
1.5mg per mL,1.0mg per mL,and 0.5mg per mL,respectively. USP28
USP28
Test solution—
Transfer about 100mg of dried Chondroitin Sulfate Sodium,accurately weighed,to a 100-mLvolumetric flask,dissolve in 30mLof water,
 USP28dilute with water to volume,and mix.
USP28dilute with water to volume,and mix.
Procedure—
Transfer 5.0mLof eachStandard solution and theTest solution to four separate titration vessels, and add about 25mLof Diluent.Use a phototrode to determine the endpoint turbidimetrically,either at 420nm,550nm,or 660nm.Stir until a steady reading is obtained.Set the instrument to zero in absorbance mode.Titrate withCetylpyridinium chloride solution.From a linear regression equation derived from the volumes ofCetylpyridinium chloride solution consumed,and the masses,in mg,of USP Chondroitin Sulfate Sodium RSin the aliquots of the respective Standard solutions,
and add about 25mLof Diluent.Use a phototrode to determine the endpoint turbidimetrically,either at 420nm,550nm,or 660nm.Stir until a steady reading is obtained.Set the instrument to zero in absorbance mode.Titrate withCetylpyridinium chloride solution.From a linear regression equation derived from the volumes ofCetylpyridinium chloride solution consumed,and the masses,in mg,of USP Chondroitin Sulfate Sodium RSin the aliquots of the respective Standard solutions, USP28determine the mass of chondroitin sulfate sodium in the aliquot of theTest solution taken.Calculate the percentage of chondroitin sulfate sodium in the portion taken by the formula:
USP28determine the mass of chondroitin sulfate sodium in the aliquot of theTest solution taken.Calculate the percentage of chondroitin sulfate sodium in the portion taken by the formula:
 found
found USP28in the aliquot of theTest solution;and Wis the weight,in mg,of Chondroitin Sulfate Sodium taken to prepare the Test solution.
USP28in the aliquot of theTest solution;and Wis the weight,in mg,of Chondroitin Sulfate Sodium taken to prepare the Test solution.
2000(M/W),
in which Mis the mass of Chondroitin Sulfate Sodium
1
Suitable cellulose acetate membranes for electrophoresis are available from Malta Chemetron SRL,Milano,Italy (www.maltachemetron.com);Fluka Chemical Corp.,Milwaukee,WI;and DiaSys Corp.,Waterbury,CT(www.diasys.com).
2
Suitable applications are available from DiaSys Corp.,Waterbury,CT(www.diasys.com);and Helena Laboratories,Beaumont,TX(www.helena.com).
Auxiliary Information—
Staff Liaison:Gabriel I.Giancaspro,Ph.D.,Senior Scientist and Latin American Specialist
Expert Committee:(DSB)Dietary Supplements:Botanicals
USP28–NF23Page 2068
Pharmacopeial Forum:Volume No.30(1)Page 197
Phone Number:1-301-816-8343