 1041
1041 BIOLOGICS
BIOLOGICS
Products such as antitoxins, antivenins, blood, blood derivatives, immune serums, immunologic diagnostic aids, toxoids, vaccines, and related articles that are produced under license in accordance with the terms of the federal Public Health Service Act (58 Stat. 682) approved July 1, 1944, as amended, have long been known as “biologics.” However, in Table III, Part F, of the Act, the term “biological products” is applied to the group of licensed products as a whole. For Pharmacopeial purposes, the term “biologics” refers to those products that must be licensed under the Act and comply with Food and Drug Regulations—Code of Federal Regulations, Title 21 Parts 600-680, pertaining to federal control of these products (other than certain diagnostic aids), as administered by the Center for Biologics Evaluation and Research or, in the case of the relevant diagnostic aids, by the Center for Devices and Radiological Health of the federal Food and Drug Administration.
Each lot of a licensed biologic is approved for distribution when it has been determined that the lot meets the specific control requirements for that product as set forth by the Office. Licensing includes approval of a specific series of production steps and in-process control tests as well as end-product specifications that must be met on a lot-by-lot basis. These can be altered only upon approval by the Center for Biologics Evaluation and Research and with the support of appropriate data demonstrating that the change will yield a final product having equal or superior safety, purity, potency, and efficacy. No lot of any licensed biological product is to be distributed by the manufacturer prior to the completion of the specified tests. Provisions generally applicable to biologic products include tests for potency, general safety, sterility, purity, water (residual moisture), pyrogens, identity, and constituent materials (Sections 610.10 to 610.15 and see
Safety Tests—Biologicals under
Biological Reactivity Tests, In Vivo  88
88
,
Sterility Tests  71
71
,
Water Determination  921
921
, and
Pyrogen Test  151
151
, as well as
Bacterial Endotoxins Test  85
85
). Constituent materials include ingredients, preservatives, diluents and adjuvants (which generally should meet compendial standards), extraneous protein in cell-culture produced vaccines (which, if other than serum-originating, is excluded) and antibiotics other than penicillin added to the production substrate of viral vaccines (for which compendial monographs on antibiotics and antibiotic substances are available). Additional specific safety tests are also required to be performed on live vaccines and certain other items. Where standard preparations are made available by the Center for Biologics Evaluation and Research (Section 610.20), such preparations are specified for comparison in potency or virulence testing. The U.S. Opacity Standard is used in estimating the bacterial concentration of certain bacterial vaccines and/or evaluating challenge cultures used in tests of them. (See also
Units of Potency in the
General Notices.)
The Pharmacopeial monographs conform to the Food and Drug Regulations in covering those aspects of identity, quality, purity, potency, and packaging and storage that are of particular interest to pharmacists and physicians responsible for the purchase, storage, and use of biologics. Revisions of the federal requirements affecting the USP monographs will be made the subjects of USP Supplements as promptly as practicable.
Vehicles and Added Substances—
Vehicles and added substances suitable for biologics are those named in the Food and Drug Regulations.
Containers for Injections—
Containers for biologics intended to be administered by injection meet the requirements for
Containers for Injections under
Injections  1
1
.
Volume in Container—
The volumes in containers of biologics intended to be administered by injection meet the requirements for
Volume in Container under
Injections  1
1
.
Labeling—
Biologics intended to be administered by injection comply with the requirements for
Labeling under
Injections  1
1
. In addition, the label on the final container for each biologic states the following: the title or proper name (the name under which the product is licensed under the Public Health Service Act); the name, address, and license number of the manufacturer; the lot number; the expiration date; and the recommended individual dose for multiple-dose containers. The package label includes all of the above, with the addition of the following: the preservative used and its amount; the number of containers, if more than one; the amount of product in the container; the recommended storage temperature; a statement, if necessary, that freezing is to be avoided; and such other information as the Food and Drug regulations may require.
Packaging and Storage—
The labeling gives the recommended storage temperature (see
General Notices). Precautions should be taken where products labeled to be stored at a temperature between 2
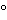
and 8
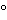
are stored in a refrigerator, in order to assure that they will not be frozen. Diluents packaged with biologics should not be frozen. Some products (as defined in Section 600.15) are to be maintained during shipment at specified temperatures.
Expiration Date—
For compendial articles the expiration date identifies the time during which the article may be expected to meet the requirements of the Pharmacopeial monograph, provided it is kept under the prescribed storage conditions. This date limits the time during which the product may be dispensed or used (see General Notices, page 1). However, for biological products, the stated date on each lot determines the dating period, which begins on the date of manufacture (Section 610.50) and beyond which the product cannot be expected beyond reasonable doubt to yield its specific results and to retain the required safety, purity, and potency (Section 300.3 (1) and (m)). Such a dating period may comprise an in-house storage period during which it is permitted to be held under prescribed conditions in the manufacturer's storage, followed by a period after issue therefrom. The individual monographs usually indicate both the latter period and (in parentheses) the permissible in-house storage period. If the product is held in the manufacturer's storage for a longer period than that indicated (in parentheses), the expiration date is set so as to reduce the dating period after issue from the manufacturer's storage by a corresponding amount.
Auxiliary Information—
Staff Liaison :
Larry N. Callahan, Ph.D., Senior Scientist
Expert Committee : (BBPP05) Biologics and Biotechnology - Proteins and Polysaccharides
USP31–NF26 Page 402
Phone Number : 1-301-816-8385

