Packaging and storage—
Preserve in well-closed containers.
Labeling—
The label states the nominal particle size range.
Identification—
A 1:10 suspension gives a violet to deep blue color with
iodine TS.
Specific rotation  781
781 :
:
not less than +41
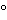
and not more than +61
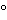
, corresponding to not less than 62.5% and not more than 91.5% of sucrose (C
12H
22O
11), calculated on the dried basis.
Test solution—
Transfer about 20 g accurately weighed, to a 200-mL volumetric flask, add 160 mL of water, shake to dissolve the sucrose, add water to volume, and mix. Pass the solubilized sucrose solution by vacuum filtration through fine filter paper.
Microbial limits  61
61 —
—
The Spheres meet the requirements of the tests for absence of
Salmonella species,
Escherichia coli,
Staphylococcus aureus, and
Pseudomonas aeruginosa, and the total aerobic microbial count does not exceed 100 cfu per g.
Loss on drying  731
731 —
—
Dry the Spheres at 105
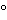
for 4 hours: the material loses not more than 4.0% of its weight.
Change to read:
Particle size 
 NF26
NF26  786
786 :
:
not less than 90% of it passes the coarser sieve size stated in the labeling; all of it passes the next coarser sieve size listed in
Table 1 of the general chapter. Not more than 10% passes the finer sieve size stated in the labeling.
(Official until July 1, 2008)
Auxiliary Information—
Staff Liaison :
Robert H. Lafaver, B.A., Scientist
Expert Committee : (EM105) Excipient Monographs 1
USP31–NF26 Page 1247
Pharmacopeial Forum : Volume No. 32(6) Page 1777
Phone Number : 1-301-816-8335

