Lactose Monohydrate
» Lactose Monohydrate is a natural disaccharide, obtained from milk, which consists of one glucose and one galactose moiety. [note—Lactose Monohydrate may be modified as to its physical characteristics. It may contain varying proportions of amorphous lactose.]
Packaging and storage—
Preserve in tight containers.
Labeling—
Where the labeling states the particle size distribution, it also indicates the d10, d50, and d90 values and the range for each. For modified Lactose Monohydrate, also label it to indicate the method of modification.
USP Reference standards  11
11 —
—
USP Lactose Monohydrate RS.
USP Sucrose RS.
USP Fructose RS.
USP Dextrose RS.
USP Lactose Monohydrate RS.
USP Sucrose RS.
USP Fructose RS.
USP Dextrose RS.
Clarity and color of solution—
A solution of 1 g in 10 mL of boiling water is clear and nearly colorless. Determine the absorbance of this solution at a wavelength of 400 nm. The absorbance divided by the path length in centimeters is not more than 0.04.
Identification—
B:
Diluent—Prepare a mixture of methanol and water (3:2).
Developing solvent—
Prepare a solution consisting of a mixture of ethylene dichloride, glacial acetic acid, methanol, and water (50:25:15:10).
Standard solution A—
Prepare a solution of USP Lactose Monohydrate RS in Diluent having a known concentration of 0.5 mg per mL.
Standard solution B—
Prepare a solution of USP Dextrose RS, USP Lactose Monohydrate RS, USP Fructose RS, and USP Sucrose RS in Diluent having a known concentration of 0.5 mg per mL for each Reference Standard.
Test solution—
Transfer about 25 mg of Lactose Monohydrate to a 50-mL volumetric flask, dissolve in and dilute with Diluent to volume, and mix.
Procedure—
Apply separately 2 µL each of Standard solution A, Standard solution B, and the Test solution to a thin-layer chromatographic plate (see Chromatography  621
621 ) coated with a 0.25-mm layer of chromatographic silica gel. Allow the spots to dry, and develop the plate in a paper-lined chromatographic chamber equilibrated with Developing solvent for about 1 hour prior to use. Allow the chromatogram to develop until the solvent front has moved about three-quarters of the length of the plate. Remove the plate from the chamber, dry in a current of warm air, and redevelop the plate in fresh Developing solvent. Remove the plate from the chamber, mark the solvent front, and dry the plate in a current of warm air. Spray the plate evenly with a solution containing 0.5 g of thymol in a mixture of 95 mL of alcohol and 5 mL of sulfuric acid. Heat the plate at 130
) coated with a 0.25-mm layer of chromatographic silica gel. Allow the spots to dry, and develop the plate in a paper-lined chromatographic chamber equilibrated with Developing solvent for about 1 hour prior to use. Allow the chromatogram to develop until the solvent front has moved about three-quarters of the length of the plate. Remove the plate from the chamber, dry in a current of warm air, and redevelop the plate in fresh Developing solvent. Remove the plate from the chamber, mark the solvent front, and dry the plate in a current of warm air. Spray the plate evenly with a solution containing 0.5 g of thymol in a mixture of 95 mL of alcohol and 5 mL of sulfuric acid. Heat the plate at 130 for 10 minutes: the principal spot obtained from the Test solution corresponds in appearance and RF value to that obtained from Standard solution A. The test is not valid unless the chromatogram obtained with Standard solution B shows four clearly discernible spots, disregarding any spots at the origin.
for 10 minutes: the principal spot obtained from the Test solution corresponds in appearance and RF value to that obtained from Standard solution A. The test is not valid unless the chromatogram obtained with Standard solution B shows four clearly discernible spots, disregarding any spots at the origin.
C:
Dissolve 250 mg in 5 mL of water. Add 3 mL of ammonium hydroxide, and heat in a water bath at 80 for 10 minutes: a red color develops.
for 10 minutes: a red color develops.
Specific rotation  781
781 —
Dissolve 10 g by heating in 80 mL of water to 50
—
Dissolve 10 g by heating in 80 mL of water to 50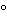 . Allow to cool, and add 0.2 mL of 6 N ammonium hydroxide. Allow to stand for 30 minutes, and dilute with water to 100 mL: the specific rotation, calculated on the anhydrous basis, determined at 20
. Allow to cool, and add 0.2 mL of 6 N ammonium hydroxide. Allow to stand for 30 minutes, and dilute with water to 100 mL: the specific rotation, calculated on the anhydrous basis, determined at 20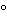 , is between +54.4
, is between +54.4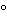 and +55.9
and +55.9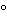 .
.
Microbial enumeration tests  61
61 and Tests for specified microorganisms
and Tests for specified microorganisms  62
62 —
The total aerobic microbial count does not exceed 100 cfu per g, the total combined molds and yeasts count does not exceed 50 cfu per g, and it meets the requirements of the test for absence of Escherichia coli.
—
The total aerobic microbial count does not exceed 100 cfu per g, the total combined molds and yeasts count does not exceed 50 cfu per g, and it meets the requirements of the test for absence of Escherichia coli.
Acidity or alkalinity—
Dissolve 6 g by heating in 25 mL of carbon dioxide-free water, cool, and add 0.3 mL of phenolphthalein TS: the solution is colorless, and not more than 0.4 mL of 0.1 N sodium hydroxide is required to produce a red color.
Loss on drying  731
731 —
Dry it at 80
—
Dry it at 80 for 2 hours: the monohydrate form loses not more than 0.5% of its weight, and the modified monohydrate form loses not more than 1.0% of its weight.
for 2 hours: the monohydrate form loses not more than 0.5% of its weight, and the modified monohydrate form loses not more than 1.0% of its weight.
Water, Method I  921
921 :
between 4.5% and 5.5%, determined on a preparation containing lactose monohydrate in a mixture of methanol and formamide (2:1).
:
between 4.5% and 5.5%, determined on a preparation containing lactose monohydrate in a mixture of methanol and formamide (2:1).
Residue on ignition  281
281 :
not more than 0.1%, determined on a specimen ignited at a temperature of 600 ± 25
:
not more than 0.1%, determined on a specimen ignited at a temperature of 600 ± 25 .
.
Heavy metals  231
231 —
Dissolve 4 g in 20 mL of warm water, add 1 mL of 0.1 N hydrochloric acid, and dilute with water to 25 mL: the limit is 5 µg per g.
—
Dissolve 4 g in 20 mL of warm water, add 1 mL of 0.1 N hydrochloric acid, and dilute with water to 25 mL: the limit is 5 µg per g.
Protein and light-absorbing impurities  851
851 —
Measure the light absorption of a 1% (w/v) solution in the range of 210 to 300 nm. The absorbance divided by the path length in centimeters is not more than 0.25 in the range of 210 to 220 nm and is not more than 0.07 in the range of 270 to 300 nm.
—
Measure the light absorption of a 1% (w/v) solution in the range of 210 to 300 nm. The absorbance divided by the path length in centimeters is not more than 0.25 in the range of 210 to 220 nm and is not more than 0.07 in the range of 270 to 300 nm.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Kevin T. Moore, Ph.D.
Scientist 1-301-816-8369 |
(EM105) Excipient Monographs 1 |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
|
| Radhakrishna S Tirumalai, Ph.D.
Senior Scientist 1-301-816-8339 |
(MSA05) Microbiology and Sterility Assurance | |
| Radhakrishna S Tirumalai, Ph.D.
Senior Scientist 1-301-816-8339 |
(MSA05) Microbiology and Sterility Assurance |
USP32–NF27 Page 1264
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.