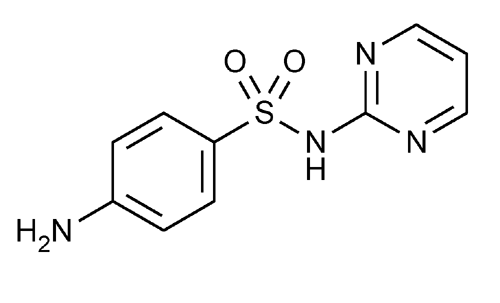
Sulfadiazine
Benzenesulfonamide, 4-amino-N-2-pyrimidinyl-.
N1-2-Pyrimidinylsulfanilamide
» Sulfadiazine contains not less than 98.0 percent and not more than 102.0 percent of C10H10N4O2S, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed, light-resistant containers.
Clarity and color of solution—
Dissolve 1 g in a mixture of 20 mL of water and 5 mL of 1 N sodium hydroxide. The solution is clear and not more deeply colored than pale yellow.
Identification—
B:
Carefully melt about 50 mg in a small test tube: a reddish brown color develops. The fumes evolved during the decomposition do not discolor moistened lead acetate test paper (distinction from sulfathiazole).
C:
Gently heat about 1 g in a small test tube until a sublimate is formed. Collect a few mg of the sublimate with a glass rod, and mix in a test tube with 1 mL of a 1 in 20 solution of resorcinol in alcohol. Add 1 mL of sulfuric acid, and mix by shaking: a deep red color appears at once. Cautiously dilute the mixture with 25 mL of ice-cold water, and add an excess of 6 N ammonium hydroxide: a blue or reddish blue color is produced.
Acidity—
Digest 2.00 g with 100 mL of water at about 70 for 5 minutes. Cool at once to room temperature, and filter. To 25.0 mL of the filtrate add 2 drops of phenolphthalein TS, and titrate with 0.10 N sodium hydroxide: not more than 0.20 mL is required to produce a pink color.
for 5 minutes. Cool at once to room temperature, and filter. To 25.0 mL of the filtrate add 2 drops of phenolphthalein TS, and titrate with 0.10 N sodium hydroxide: not more than 0.20 mL is required to produce a pink color.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 2 hours: it loses not more than 0.5% of its weight.
for 2 hours: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Selenium  291
291 :
0.003%, a 200-mg test specimen being used.
:
0.003%, a 200-mg test specimen being used.
Heavy metals, Method II  231
231 :
0.002%.
:
0.002%.
Ordinary impurities  466
466 —
—
Test solution:
8.3 mg per mL, in a mixture of toluene and dimethylformamide (2:1).
Standard solutions:
0.008, 0.041, 0.08, and 0.17 mg per mL, in a mixture of toluene and dimethylformamide (2:1).
Eluant:
a mixture of chloroform, methanol, and ammonium hydroxide (30:12:1).
Visualization:
11.
Assay—
Mobile phase—
Prepare a suitable degassed solution of water, acetonitrile, and glacial acetic acid (87:12:1).
Standard preparation—
Dissolve an accurately weighed quantity of USP Sulfadiazine RS in 0.025 N sodium hydroxide to obtain a solution having a known concentration of about 1 mg per mL.
Assay preparation—
Transfer about 100 mg of Sulfadiazine, accurately weighed, to a 100-mL volumetric flask, add 0.025 N sodium hydroxide to volume, and mix.
Chromatographic system
(see Chromatography  621
621 )—The liquid chromatograph is equipped with a 254-nm detector and a 4-mm × 30-cm column that contains packing L1. The flow rate is about 2 mL per minute. Chromatograph five replicate injections of the Standard preparation, and record the peak responses as directed for Procedure: the relative standard deviation is not more than 2.0%, and the tailing factor for sulfadiazine is not more than 1.5.
)—The liquid chromatograph is equipped with a 254-nm detector and a 4-mm × 30-cm column that contains packing L1. The flow rate is about 2 mL per minute. Chromatograph five replicate injections of the Standard preparation, and record the peak responses as directed for Procedure: the relative standard deviation is not more than 2.0%, and the tailing factor for sulfadiazine is not more than 1.5.
Procedure—
Separately inject equal volumes (about 10 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Calculate the quantity, in mg, of C10H10N4O2S in the portion of Sulfadiazine taken by the formula:
100C(rU / rS)
in which C is the concentration, in mg per mL, of USP Sulfadiazine RS in the Standard preparation; and rU and rS are the peak responses for sulfadiazine obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Behnam Davani, Ph.D., M.B.A.
Senior Scientist 1-301-816-8394 |
(MDAA05) Monograph Development-Antivirals and Antimicrobials |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3617
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.