Sterile Purified Water
H2O




 18.02
18.02
note—For microbiological guidance, see general information chapter Water for Pharmaceutical Purposes  1231
1231 .
.
» Sterile Purified Water is Purified Water sterilized and suitably packaged. It contains no antimicrobial agent.
note—Do not use Sterile Purified Water in preparations intended for parenteral administration. For such purposes use Water for Injection, Bacteriostatic Water for Injection, or Sterile Water for Injection.
Packaging and storage—
Preserve in suitable, tight containers.
Labeling—
Label it to indicate the method of preparation and that it is not for parenteral administration.
Sterility  71
71 :
meets the requirements.
:
meets the requirements.
Water conductivity  645
645 —
Perform Stage 2, Step 4 using a sufficient amount of water to perform the test. The conductivity is not more than 25 µS/cm for containers with a nominal volume of 10 mL or less at 25 ± 1
—
Perform Stage 2, Step 4 using a sufficient amount of water to perform the test. The conductivity is not more than 25 µS/cm for containers with a nominal volume of 10 mL or less at 25 ± 1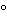 ; and not more than 5 µS/cm for containers with a nominal volume greater than 10 mL at 25 ± 1
; and not more than 5 µS/cm for containers with a nominal volume greater than 10 mL at 25 ± 1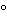 .
.
Oxidizable substances—
To 100 mL, add 10 mL of 2 N sulfuric acid, and heat to boiling. For Sterile Purified Water in containers having a fill volume of less than 50 mL, add 0.4 mL of 0.02 M potassium permanganate, and boil for 5 minutes; where the fill volume is 50 mL or more, add 0.2 mL of 0.02 M potassium permanganate, and boil for 5 minutes. If a precipitate forms, cool in an ice bath to room temperature, and pass through a sintered-glass filter: the pink color does not completely disappear.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
| Monograph | Gary E. Ritchie, M.Sc.
Scientific Fellow 1-301-816-8353 |
(PW05) Pharmaceutical Waters 05 |
| Radhakrishna S Tirumalai, Ph.D.
Senior Scientist 1-301-816-8339 |
(MSA05) Microbiology and Sterility Assurance |
USP32–NF27 Page 3872
Pharmacopeial Forum: Volume No. 31(3) Page 804