Dissolution  711
711 —
—
Medium:
water; 500 mL.
Apparatus 1:
100 rpm.
Times:
30 minutes; 60 minutes.
Mobile phase and Chromatographic system—
Prepare as directed in the Assay.
Procedure—
Inject an accurately measured volume (about 50 µL) of a filtered portion of the solution under test into the chromatograph, record the chromatogram, and measure the response for the major peak. Calculate the quantity of C
15H
10Cl
2N
2O
2 dissolved by comparison of the peak response obtained from a similarly chromatographed Standard solution having a known concentration of
USP Lorazepam RS in water.
[note—A volume of alcohol not exceeding 10% of the final volume of the Standard solution is used initially to dissolve
USP Lorazepam RS.
]
Tolerances—
The percentage of the labeled amount of C15H10Cl2N2O2 dissolved from the Tablets is not less than 60% (Q) in 30 minutes and not less than 80% (Q) in 60 minutes.
Uniformity of dosage units  905
905 :
:
meet the requirements.
Procedure for content uniformity—
Diluent, Mobile phase, and Chromatographic system—
Prepare as directed in the Assay.
Standard solution—
Prepare as directed for Standard preparation in the Assay.
Test solution—
Place 1 Tablet in a volumetric flask of appropriate size, based on the labeled quantity, in mg, of lorazepam in the Tablet, to obtain a solution having a concentration of about 0.1 mg of lorazepam per mL. Add a volume of Diluent equal to about 50% of the volume of the flask, sonicate for 10 minutes, and shake by mechanical means for 20 minutes. Dilute with Diluent to volume, mix, and centrifuge a portion of the solution for 10 minutes at 2000 rpm.
Procedure—
Separately inject equal volumes (about 20 µL) of the
Test solution and the
Standard solution into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Calculate the quantity, in mg, of lorazepam (C
15H
10Cl
2N
2O
2) in the Tablet taken by the formula:
(TC/D)(rU / rS)
in which
T is the labeled quantity, in mg, of lorazepam in the Tablet;
C is the concentration, in mg per mL, of
USP Lorazepam RS in the
Standard solution; D is the concentration, in mg per mL, of lorazepam in the
Test solution, based on the labeled quantity per Tablet and the extent of dilution; and
rU and
rS are the lorazepam peak responses obtained from the
Test solution and the
Standard solution, respectively.
Change to read:
Related compounds—
 Mobile phase—
Mobile phase—
Prepare a mixture of water, acetonitrile, and glacial acetic acid (50:50:1.2). Make adjustments if necessary (see
System Suitability under
Chromatography  621
621
).
Buffer—
Dissolve 67.7 g of sodium acetate trihydrate in 1 L of water. Adjust with glacial acetic acid to a pH of 5.0 ± 0.05.
Diluent—
Prepare a mixture of methanol and Buffer (75:25).
Standard solution—
Dissolve an accurately weighed quantity of
USP Lorazepam RS in
Diluent, and dilute quantitatively, and stepwise if necessary, to obtain a solution having a known concentration of about 0.0016 mg per mL of lorazepam.
Test solution—
Grind the number of Tablets required in order to make the total amount of lorazepam in the final composite powder about 25 mg. Accurately weigh and transfer an amount of powder equivalent to about 21.3 mg of lorazepam to a 25-mL volumetric flask. Pipet 20 mL of Diluent into the flask, and stir for 15 minutes. Do not dilute to volume. Centrifuge for 15 minutes at 2000 rpm. Pass the supernatant through a polyethersulfone membrane filter having a porosity of 0.45 µm. Quantitatively dilute the filtrate with Diluent to obtain a final solution having a known concentration of about 0.16 mg per mL of lorazepam, based on the label claim.
Chromatographic system (see Chromatography  621
621 )—
)—
The liquid chromatograph is equipped with a sample compartment chiller maintained at 4
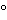
, a UV detector set at 230 nm, and a 4.6-mm × 25-cm column that contains 5-µm packing L1. The column temperature is maintained at 5
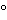
. The flow rate is about 1.0 mL per minute. Chromatograph the
Peak identification solution, record the peak responses as directed for
Procedure, and identify the peaks, using the retention times given in
Table 1.
Table 1
| Peak Identification |
Approximate
Relative
Retention Time |
Relative
Response
Factor |
Limit (%) |
| Lorazepam |
1.0 |
1.0 |
— |
| Lorazepam related compound D1 |
1.4 |
1.0 |
0.5 |
| Lorazepam related compound A*2 |
1.7 |
N/A |
N/A |
| Lorazepam related compound E3 |
1.9 |
1.3 |
0.5 |
| Lorazepam related compound C4 |
2.1 |
1.0 |
3.0 |
| Lorazepam related compound B5 |
5.5 |
1.0 |
0.1 |
| Any individual unspecified degradation product |
— |
1.0 |
0.2 |
| Total impurities |
— |
— |
4.0 |
|
*
Lorazepam related compound A is included only for peak identification purposes. It is not quantified and should not be included in the total impurities calculation.
1
6-Chloro-4-(o-chlorophenyl)-2-quinazolinecarboxylic acid.
2
7-Chloro-5-(o-chlorophenyl)-1,3-dihydro-3-acetoxy-2H-1,4-benzodiazepin-2-one.
3
6-Chloro-4-(o-chlorophenyl)-2-quinazoline methanol.
4
6-Chloro-4-(o-chlorophenyl)-2-quinazolinecarboxaldehyde.
5
2-Amino-2¢,5-dichlorobenzophenone.
|
The resolution,
R, between lorazepam related compound A and lorazepam related compound E is not less than 1.2. Chromatograph the
Standard solution, and record the peak responses as directed for
Procedure: the tailing factor for lorazepam is not more than 2.0, and the relative standard deviation for replicate injections is not more than 5% for lorazepam.
Procedure—
Separately inject equal volumes (about 20 µL) of the
Standard solution and the
Test solution into the chromatograph, collect the data for at least 50 minutes, and measure the responses for all the peaks. Calculate the percentage of each impurity in the portion of Tablets taken by the formula:
100(1/ F)(CS / CU)(ri / rS)
in which
F is the relative response factor for any given impurity found in
Table 1; CS is the concentration of lorazepam in the
Standard solution; CU is the concentration, in mg per mL, of lorazepam in the
Test solution, based on the label claim;
ri is the peak response for each impurity obtained from the
Test solution; and
rS is the peak response for lorazepam obtained from the
Standard solution. Table 1 shows the acceptance criteria for each impurity.
 1S (USP31)
1S (USP31)
Assay—
Diluent—
Prepare a mixture of methanol and water (17:3).
Mobile phase—
Prepare a filtered and degassed mixture of water, acetonitrile, and glacial acetic acid (60:40:0.4). Make adjustments if necessary (see
System Suitability under
Chromatography  621
621
).
Standard preparation—
Dissolve an accurately weighed quantity of
USP Lorazepam RS in
Diluent, and dilute quantitatively, and stepwise if necessary, with
Diluent to obtain a solution having a known concentration of about 0.10 mg per mL.
Assay preparation—
Transfer 20 Tablets to a 100-mL volumetric flask. Add about 50 mL of Diluent, sonicate for 10 minutes, and shake by mechanical means for 20 minutes. Dilute with Diluent to volume, mix, and centrifuge a portion of the solution for 10 minutes at 2000 rpm. Quantitatively dilute an accurately measured volume of the clear supernatant with Diluent to obtain a solution containing about 0.1 mg of lorazepam per mL.
Chromatographic system (see Chromatography  621
621 )—
)—
The liquid chromatograph is equipped with a 230-nm detector and a 4.6-mm × 25-cm column that contains 5-µm packing L1. The flow rate is about 1 mL per minute. Chromatograph the
Standard preparation, and record the peak responses as directed for
Procedure: the tailing factor is not more than 2.0; and the relative standard deviation is not more than 2.0%.
Procedure—
Separately inject equal volumes (about 20 µL) of the
Standard preparation and the
Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Calculate the quantity, in mg, of lorazepam (C
15H
10Cl
2N
2O
2) in each Tablet taken by the formula:
100(C / 20)(VU / V)(rU / rS)
in which
C is the concentration, in mg per mL, of
USP Lorazepam RS in the
Standard preparation; VU is the final volume, in mL, of the
Assay preparation; V is the volume, in mL, of the clear supernatant taken to prepare the
Assay preparation; and
rU and
rS are the peak responses obtained from the
Assay preparation and the
Standard preparation, respectively.


 .
.  .
.  .
.  .
.  .
.